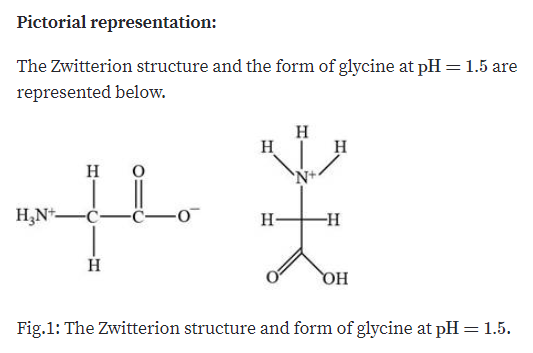
Just add up the net charge at the given pH, including charges on the amino and carboxylic acid end, and all side chains. Since 1 is less than every given pKa, we have too many protons in. This is where understanding the pI logic comes in handy. The calculation is based on the pK values of the amino acids and of the terminal amine & carboxyl. If you are asked to determine the net charge of a peptide, you can follow the same principle taught here. pI (pKa 1 + pKa 2) / 2 Isoelectric Point for Acidic and Basic Amino Acids. At this pH the the amino acid sequence forms a zwitterion. If side group has either oxygen or sulfur, protonation produces a neutral chargeĮxample 2: Determine the net charge of histidine at pH 1, 5, 9.17, and 12.Since different sources provide different values of pK values, we are averaging the isoelectric point values that are obtained using pKs from the following sources: Wikipedia. The calculation is based on the pK values of the amino acids and of the terminal amine & carboxyl group of the peptide chain. At this pH the the amino acid sequence forms a zwitterion. If side group has nitrogen, protonation produces a positive charge The isoelectric point is defined as the pH at which a particular amino acid sequence (peptide chain) bears no net electrical charge.When working with side chains, the following rule is helpful: The principle when working with amino acids having ionizable side chain is the same i.e., add up all the charges to determine the net charge. Encircle the structure of the zwitterion and calculate for the pI. At this point, glycine is in the form of a Zwitterion, having equal amounts of negative and positive charges. Draw the structures and indicate the net charge of Lysine at pH: 1.5, 7.0, 9.0, and 11.0. Notice that the net charge of glycine at pH 7 is zero. Therefore, the ionizable group will donate its hydrogen (deprotonation).Įxample 1: Determine the net charge of glycine at pH 1, 2.3, and 7. However, when pH is lower than pKa, this means that there is less hydrogen in the solution and more OH ions to be neutralized. It will thus hold on to or gain hydrogen (protonation). Therefore, there is no need to donate it to the solution to maintain a neutral acid/base balance. Relationship between pH and pkaĪnother way to look at this is that when pH is greater than the pKa (acid environment), there is a lot of hydrogen in the solution. Use the following relationship between pH and pKa to help you predict whether protonation or deprotonation will take place. The isoelectric point (pI) of an amino acid is the pH at which the molecule has an average net charge of zero and therefore does not migrate in an electric field. At the isoelectric pH NH 3 (pI 6.0), leucine is found as zwitterion. The amino group, carboxylic acid group, and side chain of an amino acid can be protonated (accept hydrogen) or deprotonated (release hydrogen) depending on the pH of the solution that the amino acid is in. The pI value can be calculated by taking the average pKa values corresponding to.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
